 Dossier Builder
Dossier BuilderDossier Builders, Reimagined with AI
Accelerate Dossier Builder development with AI-powered evidence compilation, dynamic content assembly, and automated market access documentation across global markets.
oneRay Dossier Builder at a Glance
How oneRay Dossier Builder Works
AI-powered automation for every step of the Dossier Builder development process, keeping humans in the lead.
Data Preparation & Structure
Upload or connect your data for systematic screening into oneRay Dossier Builder. Start by defining your research question. oneRay uses AI to help refine and structure your protocol, ensuring comprehensive coverage across databases.
Your data is protected at every step. oneRay helps you build a comprehensive evidence base that captures all relevant clinical, pharmacoeconomic, and outcomes data while ensuring regulatory compliance.
- AI accelerates routine compilation and formatting tasks
- Experts validate clinical judgment and strategy at every stage
- Expert review on initial outputs is mandatory, not optional
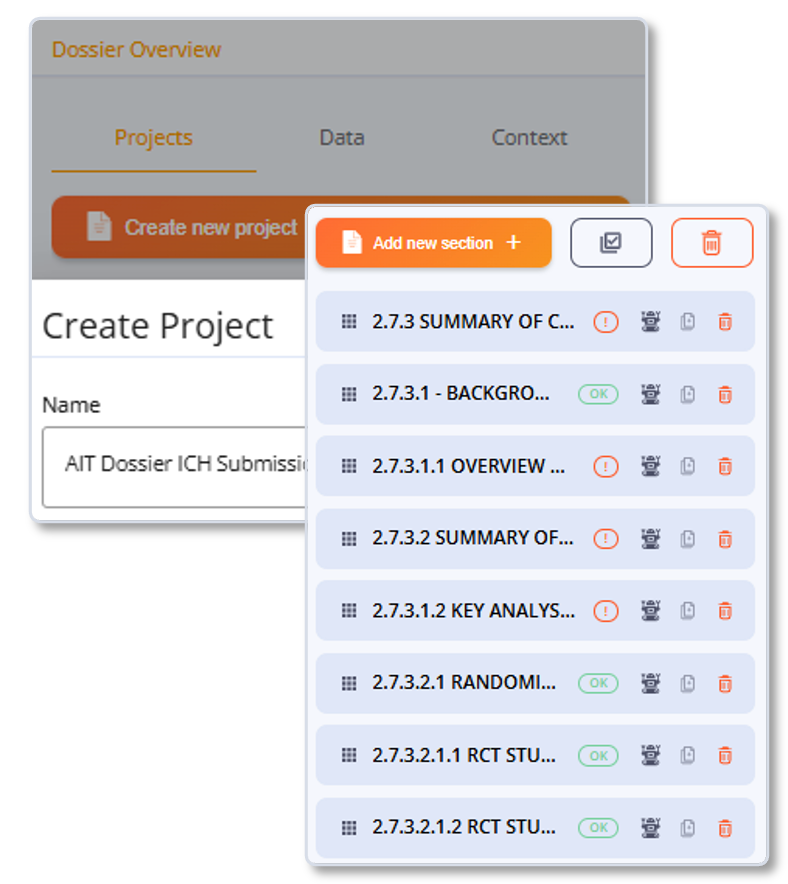
Configurations
Measure current state: document time, cost, and quality metrics of traditional dossier development. Set AI baseline by running an initial dossier to identify efficiency gains and track at scaling.
Establishing a clear baseline ensures you can quantify the impact of AI-assisted dossier development and demonstrate ROI across your organization.
- Measure current state: document time, cost, quality metrics
- Set AI baseline: run initial dossier, identify efficiency gains
- Track improvements at scaling across projects
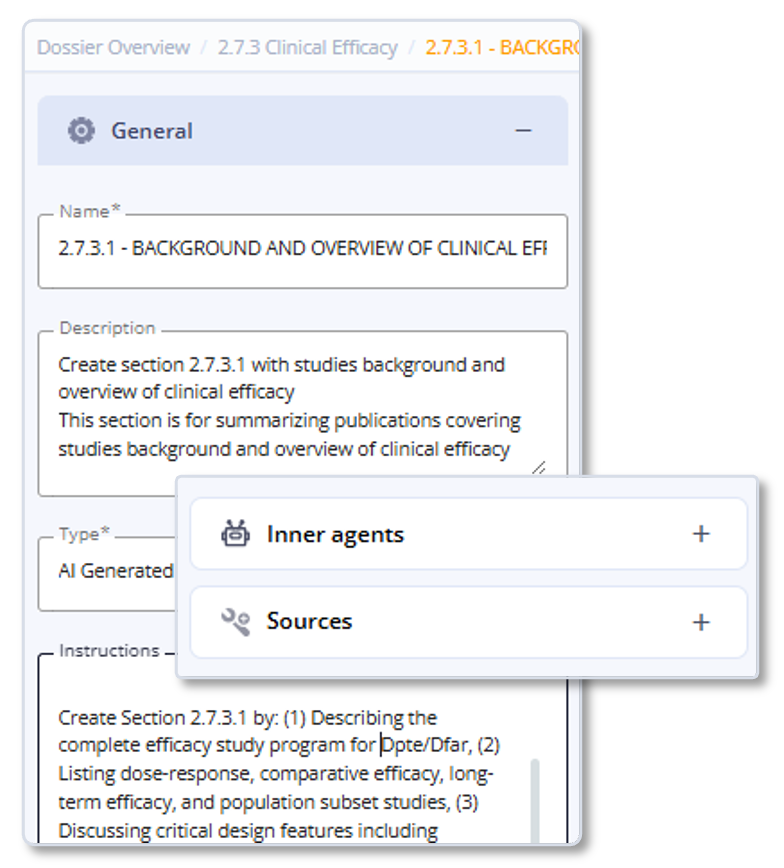
Output Review
Embed expert judgment and experience at initial AI training. Reflect emphasis, clinical intuition, regulatory strategy, and nuance in AI instructions.
Capturing implicit knowledge early ensures the AI produces outputs that reflect the depth and quality your team expects from day one.
- Embed expert judgment and experience at initial AI training
- Reflect clinical intuition and regulatory strategy in AI instructions
- Capture nuance and emphasis that differentiate your dossier
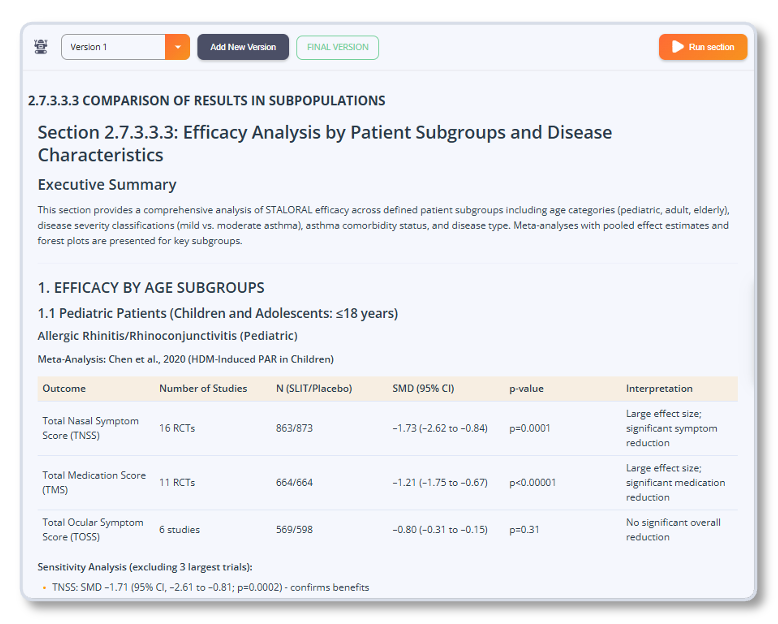
Export for Dossier Finalization
Provide full source documents and regulatory requirements upfront. Initial expert review is critical for AI training, typically requiring 1 to 3 iteration cycles.
After the baseline is set, review time drops dramatically and scale benefit emerges rapidly across all subsequent dossiers.
- Provide full source documents and regulatory requirements
- Initial expert review is critical for AI training (1–3 iteration cycles)
- After baseline is set, review time drops; scale benefit emerges rapidly

Why Choose oneRay Dossier Builder
Human-in-the-Lead
- AI accelerates routine work; experts validate clinical judgment & strategy
- Expert review on initial outputs is mandatory, not optional
Establish Clear Baseline
- Measure current state: document time, cost, quality metrics of traditional dossier development
- Set AI baseline: run initial dossier, identify efficiency gains & track at scaling
Capture In-Depth & Implicit Knowledge Early
- Embed expert judgment and experience at initial AI training
- Reflect emphasis, clinical intuition, regulatory strategy, nuance in AI instructions
Front-Load Effort, Unlock Scale
- Provide full source documents & regulatory requirements
- Initial expert review is critical for AI training (1–3 iteration cycles)
- After baseline is set, review time drops; scale benefit emerges rapidly
