 SLR
SLRSystematic Literature Reviews, Reimagined with AI
Accelerate evidence synthesis with AI-powered screening, data extraction, and automated reporting across regulatory and medical workflows.
oneRay SLR at a Glance
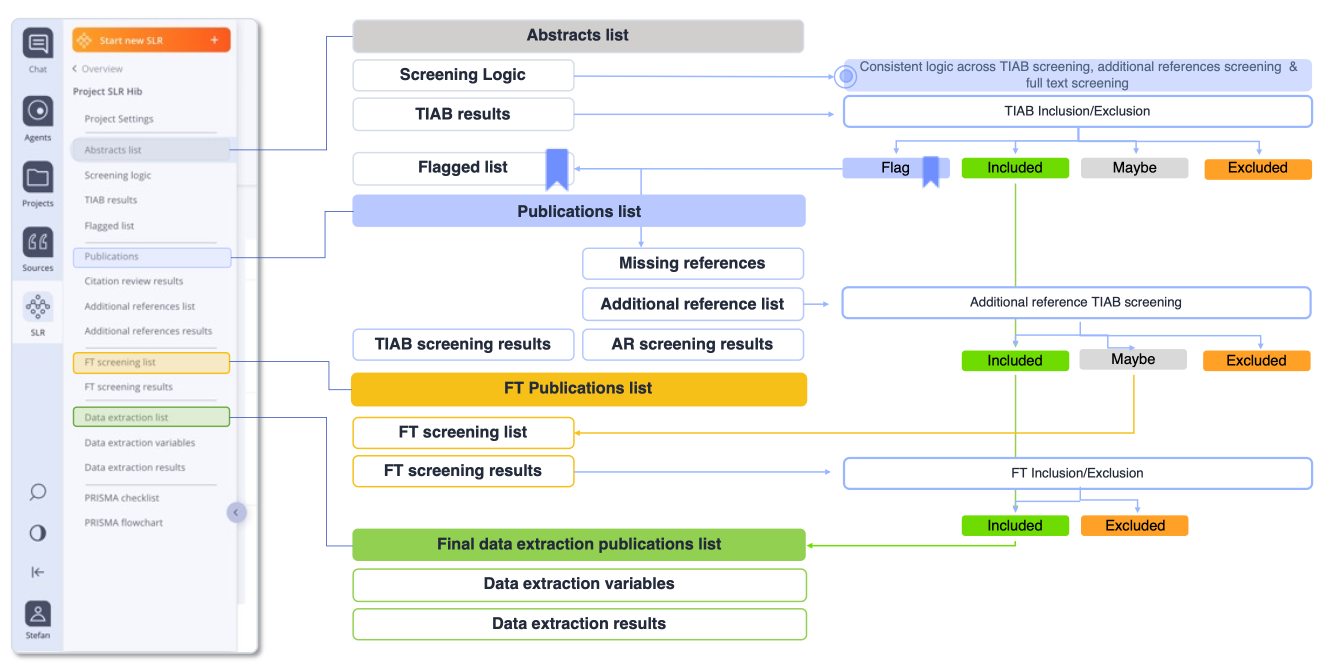
How oneRay SLR Works
AI-powered automation for every step of the systematic literature review process, keeping humans in the lead.
Define & Protect Your Research Data
Upload or connect your data for systematic screening into oneRay SLR. Start by defining your research question. oneRay uses AI to help refine and structure your protocol, ensuring comprehensive coverage across databases.
Your data is protected at every step. oneRay helps you build a robust search strategy that captures all relevant evidence while minimizing noise.
- Secure data upload and connection for systematic screening
- Automated search strategy generation across PubMed
- Multi-database query optimization
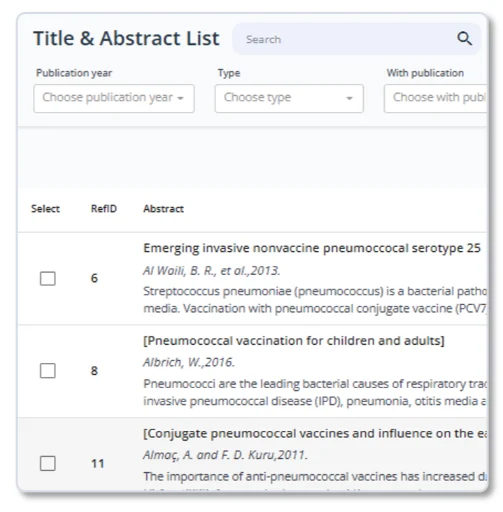
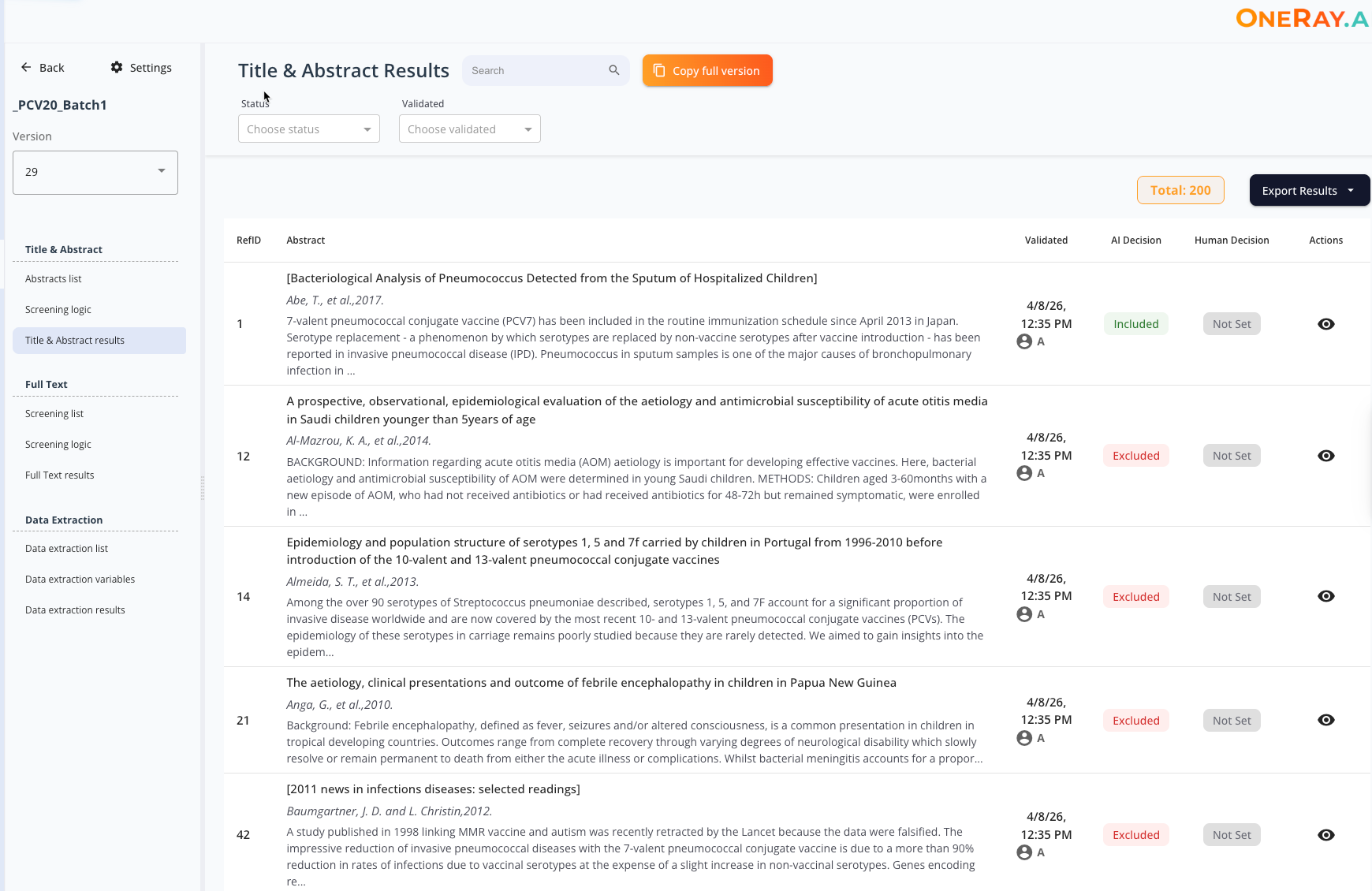
Title & Abstract Screening
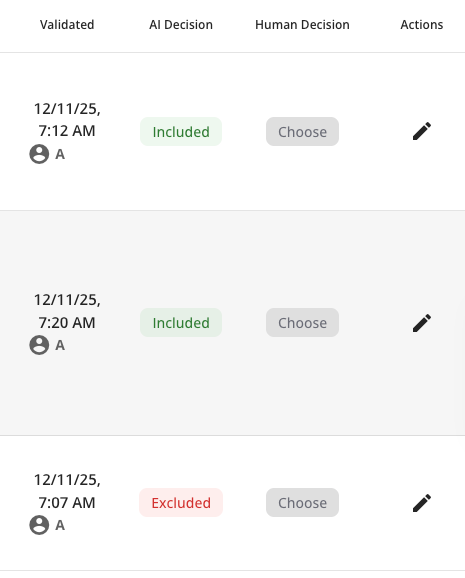
oneRay reads and evaluates every abstract against your inclusion criteria, transparently. Each decision and results come with a decision rationale, included, excluded you can review and adjust.
No more black-box screening. Every AI decision is explainable, auditable, and easy to override. Pilot test with a subset before scaling to thousands of references.
- AI screening with per-criterion scoring (include / exclude / AI Decision / Human Decision)
- Transparent rationale for every decision
- Side-by-side AI vs. Human decision columns
- Pilot testing mode for calibration
Data Extraction & Analysis
Automatically extract key data points from included studies using customizable templates. oneRay identifies study characteristics, outcomes, and risk-of-bias indicators.
From PICO elements to statistical outcomes, oneRay structures extracted data into analysis-ready formats reducing manual effort and transcription errors.
- Customizable extraction templates for any review type
- Structured data export for meta-analysis tools
- Visual summary tables and forest plot preparation
Report Generation & Export
Generate comprehensive, publication-ready reports with evidence tables, and summary of findings all formatted for regulatory or journal submission.
Whether you are submitting to a regulatory body or a peer-reviewed journal, oneRay formats your outputs to meet the required standards automatically.
- Evidence tables with customizable columns
- Export to Word, Excel, PDF, and structured XML
- Regulatory-compliant formatting (EMA, FDA, NICE)
Synthesize Evidence Across Many Sources
oneRay connects to the world’s leading scientific databases and supports your own document uploads.
Academic Papers
Search and retrieve references from the largest biomedical and scientific databases.
Clinical Trials
Access global clinical trial registries to find ongoing and completed trial evidence.
Bring Your Own Data
Upload your own reference libraries and documents for seamless integration into your review.
Why Choose oneRay SLR
Save Time
Reduce screening time by up to 75% with AI-powered automation that processes thousands of abstracts in minutes.
Ensure Quality
Maintain the highest standards with transparent AI decisions, conflict detection, and built-in quality checks.
Stay Compliant
Meet regulatory requirements with PRISMA-aligned reporting and full audit trails for every decision.
Scale Effortlessly
Handle reviews of any size from focused rapid reviews to comprehensive systematic reviews with tens of thousands of references.
Collaborate Seamlessly
Work with your team in real-time with role-based access, shared protocols, and centralized project management.
Full Transparency
Every AI decision comes with a clear rationale. Review, override, and audit every step of the process.
Ready to Accelerate Your Literature Reviews?
Join leading research teams using oneRay SLR to accelerate evidence synthesis.
